
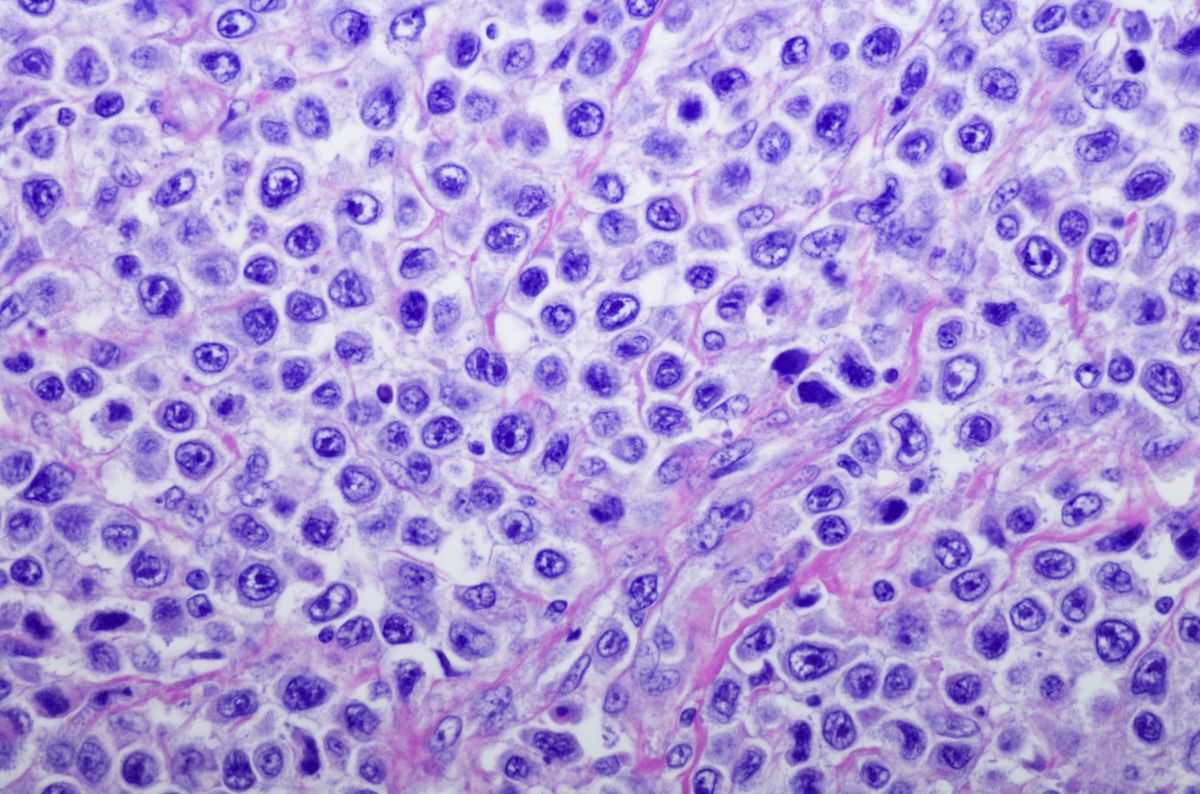
The US Food and Drug Administration (FDA) has approved nelarabine injection for the treatment of T-cell acute lymphoblastic leukemia (T-ALL) and T-cell lymphoblastic lymphoma (T-LBL).
The approval of the treatment provides patients with an alternative to a product that has historically been in shortage, according to Shorla Oncology, the manufacturer of the drug.
The company noted that in press release that the injection is a critical treatment for patients living with T-cell leukemia, particularly children with leukemia, and that it hoped the FDA’s approval addressed the clinical need in these patients.
Source: Shorla Oncology, March 2023





 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.