MDS
Myelodysplastic syndromes (MDS) are diseases of the blood cells and bone marrow. MDS are a collection of myeloid malignancies characterized by one or more peripheral blood cytopenias. Subtypes of this heterogeneous group of disorders occur when blood-forming cells in the bone marrow become abnormal. Sometimes, MDS precedes acute myeloid leukemia.
ELEMENT-MDS trial tests luspatercept vs epoetin alfa for anemia in ESA-naive, lower-risk MDS without transfusion need.
Our expert roundtable reacts to data presented at ASCO 2025 on real-world use of the agent in this setting.
Experts discuss what data do clinicians need in order to optimize their first-line use of this agent for low-risk MDS.
The document reflects the FDA’s recommendations for drug development, clinical trial designs, efficacy end points, and ...
Our expert panel reacts to the updated efficacy and safety data for luspatercept presented at ASCO 2025.
Panelists discuss how these biomarkers factor into their therapy choices and the impact of new data from ASCO 2025.
End Point Success Varies in a Phase 3 Trial of Luspatercept Plus JAK Inhibitor Therapy for MF Anemia
The combination did not meet the primary end point for RBC transfusion independence but still markedly improved anemia.Gaps in coordination and collaboration between primary care and oncology may impact long-term survival outcomes for patients.
No new safety signals were observed in this phase 3 trial which compared the combination with placebo plus azacitidine.
Bexmarilimab shows promising responses in high-risk MDS, with up to 85% ORR in the BEXMAB phase 2 study.
The comparative results from a first-of-its-kind healthcare claims database study support this conclusion.
Luspatercept shows longer survival and transfusion independence in ESA-naïve lower-risk MDS, COMMANDS trial.
Dr. Idoroenyi Amanam explains why luspatercept offers new hope for patients with low-risk myelodysplastic syndromes.
SENTI-202, a first-in-class, off-the-shelf, logic-gated CAR-NK cell therapy, is currently being evaluated in a phase 1 trial.
The phase 1/2 study's safety and efficacy data on bexmarilimab plus azacitidine for high-risk MDS are promising.
Azacitidine plus valproic acid is a safe and feasible maintenance regimen for patients with high-risk AML and MDS after HSCT.
Frontline luspatercept demonstrates superior efficacy compared with ESAs in patients with lower-risk MDS.
100% MRD-negative CR in Ph+ ALL? Dr. Catherine Lai recaps standout abstracts from an ASCO rapid oral session.
Naval Daver, MD, presented on the efficacy and safety of bexmarilimab plus standard-of-care azacitidine for MDS at ASCO 2025.
IME shows strong efficacy in LR-MDS, offering transfusion independence in heavily pretreated, ESA-ineligible patients.
Experts discuss early detection in myeloid malignancies, from current challenges to future innovations.
ADP-heptose in the intestines of older individuals accelerates the expansion of pre-leukemic blood cells.
Studies have shown that oncologists are leaving the workforce at increasing rates in association with growing workloads.
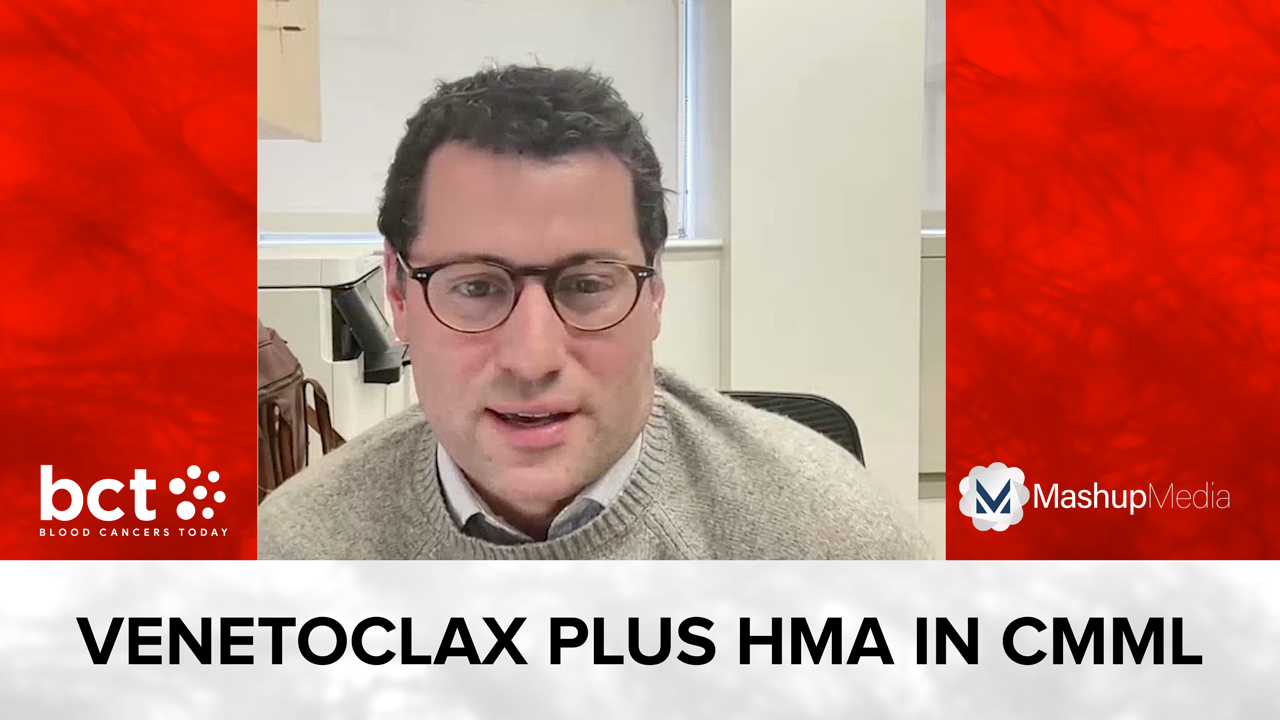
Julie Braish, MBBCh, a third-year fellow at MD Anderson, presented data on azacitidine and venetoclax in HR-MDS and CMML.
Presented preclinical model data show significant efficacy against AML cell lines with FLT3, KMT2A, NPM1, and TP53 mutations.
LucyRx’s pharmacy network includes Northwestern Medicine Specialty Pharmacy, Dana Farber Cancer Institute, and more..
Naval Daver, MD, discusses the latest advancements in the treatment of myelodysplastic syndromes
At The HemOnc Pulse Live!, experts debate key controversies in MDS care, from frontline therapy to emerging treatments.
Mice treated with CLK3 TCR9-T cells had a significantly lower tumor burden than those who received control treatments.
Anderw Brunner, MD addresses current gaps in MDS research and clinical practice and more.
The ASCO Association Board Chair has written letters to the new heads of the CMS, FDA, and NIH.
The Community Oncology Alliance outlined solutions for Congress to address systemic challenges of the U.S. healthcare system.
The investigational small molecule enzomenib has received FDA Orphan Drug and Fast Track designations in AML.
The award provides early-career researchers at NCCN Member Institutions with $150,000 in funding to support cancer research.
More than 240 meetings were held with members of Congress as part of the ASCO 10th Annual Advocacy Summit.
This agent which has newly received an ODD for MDS recalibrates the immune system to attack hematological and solid tumors.
Overall survival was also greater in the study comparing the treatments in ALL, AML, MDS, and mixed-phenotype acute leukemia.
Melody Smith, MD, MS, shares data on burnout in hematologist oncologists, why it occurs, and how to prevent it.
The approval was based on the phase 3 IMerge study, which showed improved rates of transfusion independence with imetelstat.
MRD testing before HSCT may offer insight into survival outcomes for patients with MDS/MPN.
The phase 2 prospective trial with these results also evaluated overall survival, nonrelapse mortality, and relapse rates.
Lenalidomide is indicated to treat myeloma in combination with dexamethasone and transfusion-dependent anemia due to MDS.
Nicole Grieselhuber, MD, PhD on results of SEA-CD70 treatment in patients with high-risk MDS.
Drs. Thomas LeBlanc, Jamie Koprivnikar, and Andrew Brunner give their predictions for the future of MDS management.
An expert panel delves into new data regarding the long-term efficacy of luspatercept in MDS.
An expert MDS panel discusses patient reported outcomes, transfusion dependence, and time toxicity.
This new indication for the injection alkylating agent pertains to adult or pediatric patients aged one year and older.
The COMMANDS trial evaluated frontline luspatercept in transfusion-dependent patients with lower-risk MDS and anemia.
Issues addressed include mandatory versus optional biopsy, informed consent, and safety in both adult and pediatric patients.
HMAs, either as monotherapy or in combination with venetoclax, represent the standard of care for higher-risk MDS or AML.
According to a study, patients with MDS and CMML have dismal clinical outcomes after failure of HMA therapy.
Investigational combinations for MDS appeared to be well tolerated while triplets results in high-grade toxicity.
An expert speaks on site at ASH 2024 about the health-related quality of life data from the phase 3 COMMANDS trial.
Phase 2 study results outline benefit of adding venetoclax to intensive chemo in MDS and AML patients.
Moving forward, prospective studies must validate these findings and refine risk stratification tools like the IPSS-R in MDS.
Some patients with ZRSR2-mutated CCUS had concurrent blood cancers or disorders, and others had protective co-mutation.
A discussion on the post hoc analysis on quality of life from the COMMANDS trial.
Discussing the COMMANDS trial and sequencing luspatercept in patients with myelodysplastic syndromes.
How to address ESA failure or ineligibility in patients with MDS...
Results from a real-world analysis of post-ESA treatment outcomes in patients with lower-risk myelodysplastic syndrome.
Early signs of clinical activity have been shown in a phase I/II study of tagraxofusp/decitabine in MDS and CMML.
Mature data of three immunotherapy-based hypomethylating agent combinations were presented at ASH.
High complete response rates and low early mortality rates facilitate the majority of patients to allogeneic SCT.
An ongoing phase 1 study is evaluating the safety and efficacy of danvatirsen through two substudies.
New WHO and ICC 022 guidelines propose lowering the threshold for diagnosis of CMML.
High-risk MDS is associated with poor outcomes, making the assessment of treatment response critical.
A myelofibrosis cohort showed that tapering to achieve molecular remission helped promote OS after transplant.
The design of a phase 3 trial of elritercept for transfusion-dependent anemia in lower-risk MDS was presented at ASH 2024.
Increasing evidence suggests a connection between acquired somatic mutations and inflammatory processes in myeloid neoplasms.
The clinical activity of vibecotamab in low-blast, high-risk myeloid neoplasms has been demonstrated in a phase II study.
Somatic IDH1 mutations are present in approximately 3% of patients with MDS and are associated with poor prognosis.
New research is offering new hope for these challenging case of MDS and sAML.
Ivosidenib is a promising oral therapy for patients with IDH1-mutated MDS.
Overall survival is improved in patients with SF3B1del5q-mutated MDS when lenalidomide is sequenced before luspatercept.
Findings from a phase II trial study of patients with MDS highlight the clinical benefits and safety profile of enasidenib.
Ceralasertib is a novel oral ataxia telangiectasia and Rad3-related inhibitor for patients with myelodysplastic syndromes.
The presence of genetic mutations and specific cytopenias emerged as key predictors of progression in MDS.
The phase 3 COMMANDS trial results underscore the potential longer-term clinical benefits of luspatercept in lower-risk MDS.
Phase 2 results of AZA-VEN in acute myeloid leukemia shows an efficacy benefit.
New findings underscore the potential of single-cell analyses in the unbiased unraveling of disease mechanisms in MDS.
Results from the phase 1 Combala study show the benefit of luspatercept and erythropoiesis-stimulating agents in MDS.
The phase 3 EPO-PRETAR trial demonstrates importance of early administration of ESAs in patients with MDS and anemia.
Tapotoclax was safe and may warrant consideration in combination with other therapies in MDS post-HMA failure.
ESAs had a high rate of failure among patients with lower-risk MDS and often continued treatment despite failure.
Oral azacitidine plus cedazuridine achieved pharmacokinetic equivalence to subcutaneous azacitidine in MDS and MPN.
Retrospective analysis shows that AML treatment strategies are effective for patients with MDS or CMML with NPM1 mutations.
This type of intervention produced more favorable results in patients with either hematologic or pre-hematologic relapse.
High circular RNA abundance is correlated with disease progression in patients with MDS.
The favorable effects from vamifeport pharmacologic iron restriction were enhanced by combination with luspatercept.
The model's superior engrafting of HSPCs from patients with MDS brings better replication of the disease microenvironment.
In this roundtable segment, the panel discussed imetelstat for the treatment of lower-risk MDS.
Drs. Garcia-Manero, Koprivnikar, Chedid, and LeBlanc discuss which patients with MDS should be treated with luspatercept.
Is ESA use and performance in the academic setting reflective of real-world MDS practice? Our expert panel discusses.
The panel discusses how to best implement such a monitoring system to address a widespread issue of suboptimal dosing.
Drs. Garcia-Manero, Koprivnikar, Chedid, and LeBlanc discuss dosing considerations for luspatercept for lower-risk MDS.
The panel discussed the four COMMANDS trial abstracts on luspatercept for lower-risk MDS presented at EHA 2024.
The panel led by Jamile Shammo, MD, reacts to results from a claims database study comparing the agent with ESAs.
In the next roundtable segment, the panel discusses risk stratifying patients with the IPSS-R and IPSS-M.
An expert panel discusses how to measure the MDS patient experience and whether it’s time for new endpoints in MDS.
More patients receiving luspatercept versus epoetin alfa saw improvements in erythroid, neutrophil, and platelet lineages.
More patients treated with luspatercept had improvement or resolution of neutropenia and thrombocytopenia, she said.
The expert panel discussion led by Jamile Shammo, MD, also noted differences in cell line suppression by mutational profile.
A novel multivariate model improved risk stratification in MDS compared with both the IPSS-R and IPSS-M.
The rate of abnormal cytogenetics among AA patients with MDS is comparable to or lower than the general population.
CDC database analysis suggests that MDS-related mortality has declined since 2011, though significant disparities remain.
The COMMANDS study compared the efficacy of luspatercept with epoetin alfa in ESA-naïve lower-risk MDS.
The researchers found a perfect correlation between the AI-based multiparametric flow cytometry score and the IPSS-M.
Discrepancies between the WHO and ICC classification systems for MDS can create inconsistencies in a clinical setting.
Through genomic profiling, researchers identified 18 MDS subtypes associated with clinical outcomes.
At EHA 2024, Dr. Santini shares data on luspatercept and imetelstat in patients with MDS and anemia.
Dr. Garcia-Manero discusses the latest updates from the COMMANDS trial.
Jamile Shammo, MD, leads a roundtable discussion on the effect of molecular data incorporation and goals in treatment.
Dr. David Swoboda discusses the importance of MDS World Awareness Day and what makes MDS unique compared to other diseases.
The expert panel led by Jamile Shammo, MD, tells how trial data and their clinical experiences inform their use of the agent.
A roundtable discussion, moderated by Guillermo Garcia-Manero, MD, discussed the current state of MDS treatment.
Patients who received oral HMAs had half the mean total of health care encounter days as those who received IV or SC HMAs.
A TP53 variant allele frequency threshold of 24% can inform MDS prognosis equally as well as TP53 allelic status.
The IPSS-M significantly improved MDS prognostic assessment, particularly for high-risk IPSS-R cases.
In patients with lower-risk MDS receiving luspatercept, electronic medical record alerts can help improve treatment dosing.
Dr. Garcia-Manero discussed luspatercept, a first-in-class drug for patients with low-risk MDS and anemia.
COVID-19 infection was strongly correlated with severity, blast cell percentage, and degree of dysplasia in MDS.
IDH1 inhibitor therapy significantly improved survival after HMA failure in patients with higher-risk MDS.
Luspatercept yielded high rates of durable transfusion independence in heavily-pretreated, lower-risk MDS.
Luspatercept yielded greater rates of improvement in cell lineages for patients with transfusion-dependent MDS.
COMMANDS is the first trial to perform a head-to-head comparison of luspatercept against an erythropoiesis-stimulating agent.
Enrolled patients had non-del(5q) relapsed or refractory disease and were ineligible for erythropoiesis-stimulating agents.
A retrospective study's regression analysis confirmed survival effect of cytarabine-based regimens and allogeneic HSCT.
Inherited thrombocytopenia is associated with risk for myelodysplastic syndromes and malignancies.
A shorter time to response was still observed in the azacitidine plus venetoclax cohort in this retrospective study.
In recent studies, optical genome mapping detected additional genetic aberrations in MDS compared to conventional techniques.
Most patients with MDS achieved an objective response, but limited clinical activity was observed across other blood cancers.
The two-year cumulative incidence of relapse was lower in those who achieved full donor chimerism than in those who didn’t.
“Blood Cancer Talks" hosts are joined by Dr. Stahl to discuss imetelstat and luspatercept treatment for lower-risk MDS.
An analysis of the PEGASUS and PRINCE clinical trials reported long-term efficacy of pegcetacoplan for patients with PNH.
Imetelstat led to significant improvement of red blood cell-transfusion independence rates in patients with lower-risk MDS.
Combination therapies involving PD-1 and CTLA-4 inhibitors exhibited promising activity and tolerability.
Treatment with luspatercept led to significantly less healthcare resource utilization compared with ESAs.
Dr. Raddi discusses his study on identifying predictors of response to ESA therapy in patients with lower-risk MDS.
Compared with epoetin alfa, luspatercept improved transfusion independence and hematological improvement in lower-risk MDS.
A study on this MDS subtype found that the allelic states of these mutations have implications for disease behavior.
The study compared outcomes after two conditioning types: myeloablative conditioning and reduced intensity conditioning.
There were no differences in OS among patients with NPM1-mutated myeloid neoplasms with varying bone marrow blasts.
Azacitidine plus quizartinib achieved a high ORR and a promising safety profile in patients with FLT3-Mutated MDS.
Two clinical experts offer detailed guidance on how to use iron chelation therapy in MDS care.
Multivariable analysis in a phase II study linked the supplements to an overall survival benefit in patients.
The Beat AML trial has provided practice-changing insights and allows investigators to assess the potential of novel agents.
A high percentage of MSC-like cells at MDS diagnosis carries a significantly earlier conversion of the disease to AML.
Results from a new analysis reaffirm the initial findings of the global, phase III IMerge trial.
MDS cells activate NOTCH-MYC in bone marrow mesenchymal stem cells leading to MDS disease progression.
Researchers construct a single MDS classification system which integrates the two major international systems.
The study was a noninterventional based on data collected through November 2023.
Dr. Sockel from University Hospital Carl Gustav Carus Dresden discusses recent progress and new directions in MDS management.
Elias Jabbour, MD, gives MDS highlights from EHA 2024.
Compared with epoetin alfa, luspatercept more frequently yielded improvements in anemia measures in lower-risk MDS.
A phase II study evaluated a combination of the IMM01 fusion protein with azacitidine in untreated MDS.
Achieving complete cytogenetic response results in the best overall survival in patients with MDS with abnormal cytogenetics.
A database analysis compares how much time patients spend in healthcare settings, based on receiving oral, IV, or SC HMAs.
The authors reported clinically meaningful responses of luspatercept treatment from patients enrolled in the COMMANDS trial.
ESA treatment after red blood cell transfusions was associated with worse survival compared with ESAs prior to transfusions.
The approval was based on efficacy and safety results of the randomized, placebo-controlled IMerge study.
Dr. Garcia-Manero discussed the phase II ASTREON trial on myelodysplastic syndromes at the 2024 ASCO Annual Meeting.
Early detection and prevention are particularly important in leukemia, Dr. Garcia-Manero explained.
Palliative care facilitates communication and helps with symptom management, the researchers noted.
Dr. Colla describes her current research at MD Anderson, such as developing new therapeutic approaches to MDS.
The average rate of progression for CHIP to MDS or AML is about 1% per year, Dr. Patel explained.
A drug approval specific to CMML would catalyze a surge of interest in the hematology-oncology community.
The Leukemia and Lymphoma Society's CMML Initiative hopes for dedicated CMML trials and CMML-directed therapies.
A significant number of patients receiving ivosidenib had reduced levels of voriconazole and posaconazole.
The case study included four patients with CMML who had KRAS mutations and a multitude of autoimmune diseases.
The study also explored the role of this combination in transitioning patients to HSCT.
An FDA committee voted that the benefits of imetelstat outweigh the risks for the treatment of anemia in lower-risk MDS.
UM171 promotes degradation of the CoREST1 complex and reduces levels of chromatin-bound MYC.
The CHMP of the European Medicines Agency recommended approval of luspatercept for transfusion-dependent anemia due to MDS.
Dr. Patel and the hosts discuss the TRANSFORM-1 study, MANIFEST-2, AUGMENT-101, and more.
Gilead said that it "will not pursue further development of magrolimab in hematologic cancers."
Oral decitabine plus cedazuridine is a safe and effective alternative to intravenous decitabine.
This episode of The HemOnc Pulse features Dr. Shastri, a physician-scientist, who discusses latest news in MDS.
Sanjay Patel, MD, discusses a study on spatial mapping of human hematopoiesis using bone marrow tissue.
Complete response was achieved in 14 patients (22%) in the sabatolimab group, versus 11 patients (18%) in the placebo group.
The panel shares their outlook for the future of MDS research and treatment.
Dr. Tanaka outlines a biomarker study that observed reduced inflammation in patients with MDS treated with luspatercept.
The panel discussed a study that assessed quality of life and PROs in patients receiving luspatercept for MDS.
The panel discusses whether starting treatment with luspatercept sooner in patients with MDS is beneficial.
Dr. Shammo shares her reaction to a study that assessed mutational burden and impact on primary outcomes in COMMANDS.
Dr. Safah outlines the findings from the full analysis of the COMMANDS trial.
The panel discusses the evolution of MDS therapy.
Dr. Safah gave an overview of myelodysplastic syndromes.
Dr. Venugopal offers her thoughts on the investigational drug KER-050 in lower-risk myelodysplastic syndromes.
The panel discusses forward-looking thoughts for the treatment of low-risk myelodysplastic syndromes.
The panel talks about the use of transplant for patients with low-risk myelodysplastic syndromes.
The panel discusses other treatments undergoing research for MDS, including imetelstat and KER-050.
The panel shared their thoughts on real-world data of luspatercept presented at ASH 2023.
The panel addresses continued MDS research needs.
The panel discusses appropriate treatment selection based on patient mutation status and comorbidities.
The panel shares their thoughts on the full analysis of the COMMANDS study.
The panel discussed the current slate of treatment options for patients with low-risk MDS.
Dr. Schienberg discussed luspatercept in MDS, selinexor in AML, and the difficulty of randomizing trials.
The study compared somatic mutations and variant allele frequencies in paired peripheral blood and bone marrow samples.
Dr. Guillermo Garcia-Manero outlined the final data update on luspatercept in patients with MDS in the COMMANDS trial.
Investigators reviewed cladribine in combination with low dose cytarabine (LDAC) and venetoclax.
In this video interview, Dr. Swoboda talks MDS highlights from ASH 2023, including the COMMANDS and IMerge trial.
Targeted next-generation sequencing of the full UBA1 locus was used to profile diagnostic and treatment-naïve samples.
Patients in the COMMANDS study were randomly assigned to luspatercept or epoetin alfa.
Of the patients included in the study, 67.6% were not transfusion dependent before initiating luspatercept.
OS was still inversely proportional to the IPSS-M, but the molecular risk stratification had no additional prognostic power.
Data differentiated the mechanisms of action of luspatercept from epoetin alfa in patients with transfusion-dependent LR-MDS.
Baseline mutational burden impacted response to erythropoiesis-stimulating agents in naïve patients with lower-risk MDS.
Venetoclax plus azacitidine was well tolerated and achieved favorable response rates in higher-risk myelodysplastic syndrome.
Researchers studied patients with LR-MDS who had received an erythropoiesis-stimulating agent as frontline therapy.
Amer Zeidan, MBBS, MHS, reflects on the COMMANDS study that compared luspatercept with epoetin alfa in MDS.
Rami Komrokji, MD, discussed the impact of luspatercept on the genomic landscape in patients with lower-risk MDS.
The study determined the maximum tolerated dose of CPX-351, but further studies are needed to evaluate its activity.
The study evaluated mesenchymal cell molecular features and sought modifications that might affect MDS.
Guillermo Garcia-Manero, MD, discusses contemporary issues regarding the management of LR-MDS.
Clinicians weigh in on AI advances in hematological diagnoses, prognoses, and treatment.
Iron may participate in the pathophysiology of MDS by increasing genomic instability and altering the microenvironment.
RNA sequencing showed decreased gene expression levels of the NF-κB target genes IL1β and IER3 after emavusertib treatment.
Red blood cell transfusion independence (RBC-TI) ≥8 weeks during weeks 1-24 of treatment was associated with improved OS.
Factors “significantly associated with overall survival" include older age, male sex, monosomal karyotype, and more.
The October 24, 2023, approval was based on trial AG120-C-001 that evaluated the safety of ivosidenib.
The study enrolled 214 participants, including 111 patients with 39 different RUNX1 variants from 45 unrelated families.
Anne Sophie Kubasch, MD, of Leipzig University, speaks on the importance of MDS World Awareness Day.
Dr. Garcia-Manero said he hopes to find “better treatments” and even “one day a cure for this condition.”
Researchers found that 16% of the patients analyzed had pre-existing autoimmune disease.
Across the entire study population, the two most frequent U2AF1 variants were S34F (n=97) and Q157P (n=46).
The Marketing Authorization Application is based on results from the IMerge phase III trial.
The proof-of-concept study showed that noninvasive hemoglobin measurement is “feasible,” but there were several ...
One aim is to evaluate the efficacy of oral decitabine/cedazuridine and magrolimab in intermediate to HR MDS.
Joseph Khoury, MD; Sanam Loghavi, MD; and Chadi Nabhan, MD, MBA, FACP, discuss the MDS classification systems.
Dr. Garcia-Manero discusses key data from the phase III COMMANDS trial.
Dr. Komrokji speaks about what might follow the recent FDA approval of luspatercept for certain patients with MDS.
Researchers sought to differentiate the mechanism of action of luspatercept from epoetin alfa.
Dr. Hasserjian discusses a presentation about the classification system that he gave at the SOHO Annual Meeting.
A research team sought in their study to determine if the IPSS-M could also apply to CMML.
"There's been a lot of focus on classifications and mutations: they're coming with a lot of force," Dr. Scheinberg said.
Dr. Platzbecker joins Chadi Nabhan, MD, MBA, FACP, to discuss MDS at the Eleventh Annual Meeting of SOHO.
Guillermo Garcia-Manero, MD, joins Chadi Nabhan, MD, MBA, FACP, to discuss the COMMANDS trial at the SOHO Annual Meeting.
Allogeneic HSCT can yield favorable outcomes in treating patients with therapy-related MDS.
A presentation by Dr. Rami Komrokji at the Eleventh SOHO Annual Meeting gave additional insights into the COMMANDS trial.
Luspatercept is highly effective in treating ESA-naïve patients with LR-MDS.
Response rates were high, and OS was improved in patients receiving venetoclax, although not statistically significant.
The expanded indication to the frontline setting is based on interim results from the pivotal phase III COMMANDS trial.
Guillermo Garcia-Manero, MD, the 2023 SOHO President-Elect, shares his hopes and goals for the coming years.
The study's authors found evidence showing that "heme might serve as a rheostat directing cells to live or die."
Gilead Sciences, Inc, the manufacturer of the drug, recommends discontinuing magrolimab treatment in patients with MDS.
The single-center phase I/II study evaluated the triplet combination after hypomethylating agent failure.
From Houston, Texas, to Beirut, Lebanon, the SOHO global community continues to grow thanks to its Ambassador Program.
The FDA has removed the partial clinical hold on the TakeAim Leukemia phase I/II study of emavusertib.
The study compared luspatercept with epoetin alfa in patients with lower-risk MDS who were naive to ESA treatment.
An NDA for the telomerase inhibitor imetelstat has been submitted to the FDA.
Dr. Winer discusses emavusertib, an orally bioavailable, reversible inhibitor of IRAK4.
Dr. Venugopal, who was not involved in the study, discusses the results and implications of the phase III trial.
The median OS was 13.3 months in patients receiving CPX-351, while it was 11.4 months in those receiving FLAG-Ida.
In the 84 patients with MDS who were identified as vulnerable, 88% had difficulty with prolonged physical activity.
The investigators replicated the statistical plan of the MEDALIST trial to evaluate luspatercept outside of a clinical trial.
KER-050 is a modified activin receptor type IIA ligand trap designed to inhibit select TGF-β superfamily ligands.
The study evaluated imetelstat versus placebo in patients with lower-risk MDS who were highly transfusion dependent.
Dr. Stéphane De Botton discusses his EHA abstract on tamibarotene-based therapy in MDS/AML.
Dr. Garcia-Manero presented results from the phase III COMMANDS trial during the 2023 ASCO Meeting.
Luspatercept-aamt demonstrated “favorable outcomes” compared with epoetin alfa across common mutations in MDS.
Most patients (82%) had a low transfusion burden at baseline.
A subset of TP53-mutated myeloid neoplasms, such as MDS, are preceded by TP53-mutated CCUS.
Nearly one-third (29%) of patients were upstaged from their IPSS-R score by the IPSS-M.
Dr. Garcia-Manero presented information about a phase Ib study of IRAK1/4 inhibition at the 2023 ASCO Annual Meeting.
In this video, Dr. Garcia-Manero discusses the study's primary endpoint, its implications for patients, and more.
Dr. Zeidan speaks about the rationale behind the trial, the implications of its results, and the next steps.
The field of hematologic oncology is now contending with reconciling the proposed rival WHO and ICC systems.
The Type II Variation Application was accepted by the European Medicines Agency (EMA) as well.
Dr. Zeidan discusses a patient who shaped his career path, the pressing questions in MDS and AML, and more.
Certain cellular pathways were upregulated in patients with myelodysplastic syndromes (MDS) who responded to immunotherapy.
Researchers retrospectively screened patients with VEXAS for clonal hematopoiesis in their peripheral blood.
The study assessed “patterns of consideration” for HSCT and identified factors associated with transplant referral rates.
The study showed starting statin therapy at MDS diagnosis was associated with a 38% improvement in OS.
Novel targets are under investigation in several types of hematologic malignancies.
The ICC and WHO both “proposed significant changes” to the diagnostic criteria for MDS in 2022.
In patients with TP53 mutations, 40% achieved a complete response, with a median OS of 16.3 months.
The three-year OS rate was 91.3% for patients who were MRD negative, while it was 66.4% for those who were MRD positive.
The novel two-stage diagnostic classifier was based on mutational profiles in 18 genes.
Post-treatment clone size significantly correlated with IWG responses, except in the case of DDX41-mutated clones.
The safety profile of eprenetapopt and venetoclax plus azacitidine was acceptable, according to this phase 1 study.
The EC approval of luspatercept was based on results from the phase II BEYOND study.
Treatment of LR-MDS remains an unmet need and goals of therapy should extend beyond transfusion independence.
MDS/AML are among independent predictors of COVID-19 treatment failure.
Oxidized mitochondrial DNA can “prime” MDS HSPCs for inflammasome activation.
Low-risk MDS can have high-risk features, but current MDS prognostic scoring systems may not capture key risk variables.
While the fifth edition “softened” the boundary between MDS and AML, it retains the 20% blast cutoff to define AML.
Exposure to thalidomide analogs is associated with TP53 mutations in patients with therapy-related myeloid neoplasms.
Luspatercept reduced RBC transfusion units and visits in patients with lower-risk MDS with ring sideroblasts.
A panel recently made a consensus proposal to revise the IWG response criteria for higher-risk MDS.
The U.S. FDA granted Fast Track Designation to tamibarotene, an oral selective RARα agonist, for higher-risk MDS.
Sanam Loghavi, MD, chats with The HemOnc Pulse host Chadi Nabhan, MD, MBA, FACP, about why 2022 was a major year for MDS.
A participant’s death led to a voluntary pause of a phase I/II trial evaluating MGTA-117 in relapsed/refractory AML and ...
Long-term safety data for luspatercept showed no new safety signals in lower-risk MDS.
Venetoclax plus azacitidine provided “clinically meaningful benefits” in R/R/ MDS post-HMA failure.
Researchers have developed and externally validated a clinical-molecular prognostic model for risk stratification in MDS.
Patients with MDS who received active therapy soon after their diagnosis had better outcomes than those who did not.
DocMatter members weigh in on the use of venetoclax in patients with higher-risk myelodysplastic syndromes.
Survival rates for MDS/MPN overlap syndromes vary significantly by clinical entity.
A phase III trial confirmed the efficacy of lenalidomide in transfusion-dependent MDS patients.
MDS patients with del5q and one additional non-del20q cytogenetic abnormality are at higher risk.
Researchers formulated an optimal dosing schedule for the oral-fixed dose combination of decitabine and cedazuridine in MDS.
Patients with MDS or AML experienced a reduction in the allele frequency of TP53 mutations in as early as cycle three.
Blast quantification modalities vary substantially and could significantly impact treatment decisions in patients with MDS.
Phillip Scheinberg, MD, talk about CHIP and the IPSS-M in MDS with Chadi Nabhan, MD, host of The HemOnc Pulse, during ASH.
Roger Lyons, MD, FACP, discusses challenges and future directions in treating patients with higher-risk MDS.
A recent study evaluated patients with rare myeloid neoplasms who had NPM1 mutations.
The IPSS-M Risk Stratification Model led to reclassification of almost half of patients with hypoplastic MDS.
The number of cells with chromosomal abnormalities was significantly associated with clinical features and outcomes in MDS.
Most patients with lower-risk MDS who had low transfusion burden before luspatercept achieved transfusion independence.
Roger Lyons, MD, FACP, discusses a real-world retrospective observational study of treatment and survival in MDS.
SX-682 was effective and well tolerated at a dose of 200 mg twice daily in patients with MDS.
A recent study evaluated the safety profile of deferiprone in patients with myelodysplastic syndromes (MDS).
A new machine-learning model based exclusively on clinical variables high prognostic accuracy for predicting MDS outcomes.
Patients with MDS or TP53-mutated acute leukemias who underwent HSCT had a one-year OS rate of 48.4%.
Venetoclax plus azacitidine maintenance after allogeneic HSCT led to a one-year OS rate of 70% in patients with AML/MDS.
More than one-third of patients with MDS and their caregivers had “concerning levels of depression and/or anxiety."
Patients aged 80 years and older and the lowest-risk patients with MDS are “more likely to die from competing causes."
A CR with partial hematologic recovery leads to "similar survival and duration on therapy” as a CR in patients with MDS.
Decitabine did not improve event-free survival over hydroxyurea in patients with advanced myeloproliferative CMML.
Eprenetapopt and azacitidine after allogeneic HSCT was well tolerated in patients with TP53-mutated AML and MDS.
Venetoclax and azacitidine in R/R MDS demonstrated a manageable safety profile with meaningful clinical benefits...
Further development of BI 836858 was discontinued after it failed to elicit a response in transfusion-dependent.
Azacitidine may be a treatment option for patients with SAID associated with MDS and CMML.
A prospective study failed to confirm the general predictive value of a biomarker-driven prediction model.
HLA-matched sibling donors are the preferred transplant choice for MDS.
A study showed "unexpected" results related to race and MDS survival...
Nearly a third (30%) of patients with higher-risk MDS have an overexpression of the RARA gene...
The classification process is mainly based on two processes, class discovery and class prediction...
Phillip Scheinberg, MD, PhD, discusses myelodysplastic syndromes at the 2022 SOHO Annual Meeting...
ASTX727 is an oral fixed-dose combination of HMA decitabine and cytidine deaminase inhibitor cedazuridine...
Blood Cancers Today interviewed Dr. Diez-Campelo after her presentation titled, “Treatment of Lower Risk MDS.”
Researchers retrospectively assessed 135 untreated patients with MDS who later developed HMA-F...
Researchers investigated its use in patients undergoing mismatched donor HSCT with post-transplant cyclophosphamide...
Lionel Adès, MD, PhD, spoke with Blood Cancers Today about the session he presented at the 2022 SOHO Annual Meeting...
The outcomes of AML transformation after HMA-failure MDS remain extremely poor...
Azacitidine is the current standard of care, and magrolimab is a monoclonal antibody blocking CD47 expression...
During the 2022 SOHO Annual Meeting, Jacqueline S. Garcia, MD, discussed immunotherapies for myelodysplastic syndromes...
There was no significant difference between patients treated with azacitidine on a five-day or a five-plus-two-day schedule..
Dr. Garcia-Manero talks about what a “total approach” to MDS means to him...
The number of HSCTs performed for MDS is increasing, yet HSCT remains under-utilized for MDS...
Guillermo Garcia-Manero, MD, was honored during the society’s 10th annual meeting in Houston, Texas...
The study sought to understand the characteristics and molecular profile of h-MDS in comparison with n-MDS...
Guillermo Garcia-Manero, MD, gives his perspective.
Alexandre Bazinet, MD, and colleagues conducted the single-center, dose-expansion phase I/II trial...
It led to an overall response rate of 74% in patients with newly diagnosed IDH2-mutated myelodysplastic syndrome.
Pathogenic or likely pathogenic germline variants are found in around a fifth of patients with MDS diagnosed at or before 40.
The results of the trial were presented during the 2022 ESMO Congress by Chunkang Chang, MD, PhD.
The FDA granted Orphan Drug Designation to tamibarotene for the treatment of MDS in February 2022.
The researchers analyzed data from previously untreated adult patients with low- or intermediate-risk MDS.
The study included patients who had anemia due to lower-risk MDS with ring sideroblasts and received regular RBC transfusions
A phase II, multicenter, open-label trial found that eprenetapopt plus azacitidine was well tolerated and safe.
The trial examined selinexor's effect on treatment outcomes in patients with AML and high-risk MDS.
The global, phase III PANTHER trial investigated the comparison in higher-risk MDS, higher-risk CMML, and AML.
Guillermo Garcia-Manero, MD, discusses where the field of hematologic oncology is headed.
Dr. Garcia-Manero discusses research from the MDS/AML Moon and what he’s looking forward to as President-Elect of SOHO.
The MDS/AML Moon Shot® program supports research projects that would be difficult to obtain traditional funding for.
A retrospective study showed that response rate and survival outcome are still modest for high-risk patients.
The study analyzed 70 transplant-ineligible patients with high-risk MDS and MDS/AML with less than 30% of myeloblasts.
The new IPSS-Molecular model generates a unique risk score for individual patients.
Researchers reviewed the FDA Adverse Event Reporting System and the EudraVigilance database for adverse event reports.
Treosulfan demonstrated a significant improvement in EFS over RIC busulfan with a clinically meaningful benefit.
HMAs are currently used as a first-line treatment for patients with MDS, but their mechanism of action is not clear.
The presence of TP53-mutated MDS and AML with CK points to a single aggressive disease.
TP53-mutated AML and MDS-EB do not differ with respect to their clinical and molecular characteristics or survival outcomes.
Study finds BM CH was present in nearly two-thirds of BPDCN patients and particularly prevalent in elderly patients.
The FDA and Finnish Medicines Agency cleared the investigational new drug application for bexmarilimab.
Dr. Scheinberg talks about how his father influenced his career and how he built an advanced hematology program in Brazil.
The phase I/II study will assess the safety and tolerability of the drug.
The hold was initiated after a reported death of a trial participant.
The FDA has put a partial hold on a clinical trial of emavusertib after the death of a patient.
The hold follows reports of a discrepancy in suspected unexpected serious adverse events between treatment arms.
Editor-in-Chief Sagar Lonial, MD, FACP, introduces the new publication from SOHO.
At SOHO 2021, Susan O'Brien, MD, and Hagop Kantarjian, MD, addressed trends in hematologic malignancies.
Jesus D. Gonzalez-Lugo, MD, and colleagues, presented the findings at the 2021 ASH Annual Meeting.
Results from the phase II IDIOME trial suggest that ivosidenib had good tolerability and significant efficacy.
These findings were shared at the 2021 ASH Annual Meeting by investigator Elsa Bernard, PhD.
A study evaluated factors associated with intense end-of-life care among patients younger than 40 with blood cancer.
Advertisement
Blood Cancers Today delivers the latest news, education, and information relevant to hematologic oncology patients and practices.
Sign up to receive Blood Cancers Today eNewsletters:
Advertisement






































































































































































































 © 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.
© 2025 Mashup Media, LLC, a Formedics Property. All Rights Reserved.